 This test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation. §263a, that meet the requirements to perform moderate, high, or waived complexity tests. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. This test is authorized for use with direct anterior nasal (nares) swab samples from individuals without symptoms or other epidemiological reasons to suspect COVID-19, when tested twice over three days with at least 36 hours between tests. The BinaxNOW COVID-19 Ag 2 Card is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 in direct anterior nasal (nares) swab samples from COVID-19 symptomatic individuals tested twice over three days with at least 36 hours between tests within the first seven days of symptom onset. 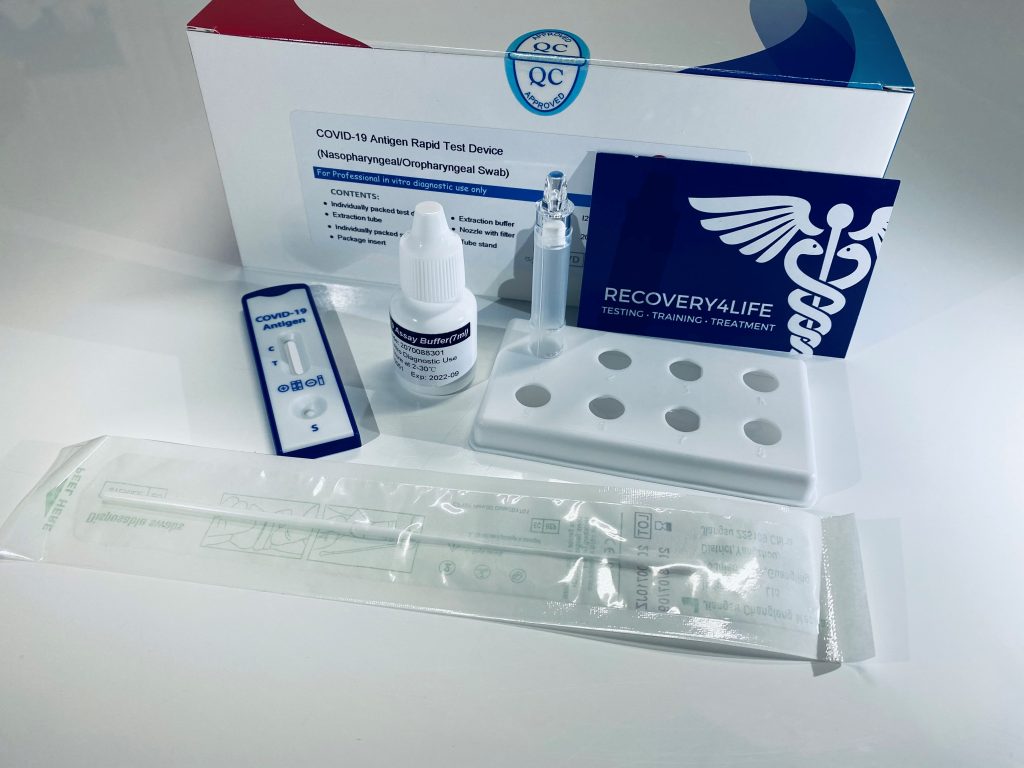
This test is authorized for non-prescription home use with self-collected direct anterior nasal (nares) swab samples from individuals aged 15 years or older or adult collected anterior nasal swab samples from individuals aged two years or older. The BinaxNOW™ COVID-19 Antigen Self Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 from individuals with or without symptoms or other epidemiological reasons to suspect COVID-19 infection when tested twice over three days with at least 36 hours between tests. No more lines, no more wait times, no more barriers and no more inconveniences. This combination will help attack the pandemic on critical fronts – speed, simplicity, affordability, access and reliability. You can now access our BinaxNOW test in 3 ways: at your local retailer over the counter self-test, or proctored at-home or from your healthcare professional. Now, with BinaxNOW authorized for over the counter for frequent asymptomatic use, we are making testing directly available for fast results, when and where you need it. The BinaxNOW COVID-19 Self Test card is identical to the professional-use test card, used since August 2020, and is the most studied and widely available rapid antigen test and is now available as a Self Test. ANSWERS, WHEN AND WHERE YOU NEED THEM.Įven while vaccines are rolling out, COVID-19 testing will remain crucial to helping us all return to normal as we begin to engage in everyday life once again. Tests have been available for purchase at chemists and supermarkets since November, with many sold in packs of two, five, 10 or more.NO MORE BARRIERS. Rapid antigen testing kits cost roughly $10-20 per single test. “These are really a useful additional layer of screening that we should certainly be using, but making sure we understand that the performance is not exactly the same as those laboratory-based tests.” Cost “If they’ve approved it, and it is available, it’s a test I would endorse.”īut Griffin was quick to point out that rapid antigen tests don’t replace PCR tests, particularly for people with symptoms. 
“I’d recommend anything approved by the TGA – we’ve seen throughout the pandemic, with regards to testing and vaccines, that they are incredibly rigorous. InnoScreen Covid-19 Antigen Rapid Test (Nasal Swab)Įcotest Covid-19 Antigen Saliva Test kit (saliva)ĬareStart Covid-19 Antigen Home Test (Nasal Swab)Īlthough the varying degrees of clinical sensitivity are listed by the TGA, the University of Queensland infectious disease physician, associate professor Paul Griffin said any test approved by the TGA was worth using. Roche Sars-CoV-2 Antigen Self Test (Nasal Swab) 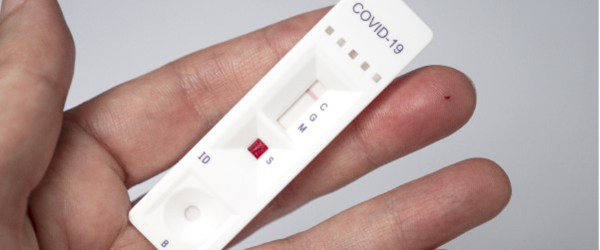
Testsealabs Covid-19 Antigen Test Cassette (Nasal Swab)Īnd finally, the tests rated as “acceptable sensitivity” are: RightSign Covid-19 Antigen Rapid Test Cassette (Nasal Swab) My Covid Test Antigen Rapid Test (Oral Fluid) The following have been given the “high sensitivity” label:Īll Test Covid-19 Antigen Rapid Test (Oral Fluid) Panbio Covid-19 Antigen Self-Test (Nasal Swab) OnSite Covid-19 Ag Self Test (Nasal Swab) Lyher Novel Coronavirus Antigen Test Kit (Nasal Swab) There are only five kits that have been given the “very high sensitivity” label:Īll Test Sars-CoV-2 Antigen Rapid Test (Nasal Swab) 
The TGA sets the minimum clinical sensitivity a testing kit must achieve before it is approved at 80%, which it labels as “acceptable sensitivity”.Ī kit with 90% PPA is labeled “high sensitivity” and a kit that records clinical sensitivity greater than 95% is considered to have “very high sensitivity”. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
